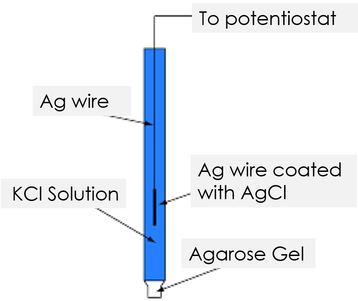
As a commercial silver-silver chloride reference electrode can cost up to $100, our group decided to construct a homemade one by ourselves.
First, we prepared the autopipettor tip with gel by mixing 1.4g agarose, 100 ml water and 5.0g KNO3, and heat the solution to boiling with constant stirring.
After preparing the solution, we placed the autopipettor tip upright in the solution and allow it to cool and gel for 24 hours.
|
Next, we prepared the silver wire with silver chloride coating by dipping a fine silver wire in the bleach solution for 10 minutes until the presence of a dark deposit on the silver wire. OCl- ion and Cl- ion in the bleach solution react with silver to form silver chloride, which is shown in the equation below. |
Finally, we filled the autopipettor tip with 3.0 M KCl solution, and inserted the wire into the tip. We used the parafilm to both prevent the evaporation of the KCl solution and stablised the silver wire.
Testing of the Ag/AgCl reference electrode
Our group tested our homemade Ag/AgCl reference electrode against a commercial one in 3.0 M KCl solution. The potential difference obtained was 2.2 mV, which is within the acceptable range of 20 mV. Hence, our homemade Ag/AgCl reference electrode can function properly.